
Hybridization: sp3d2 hybrid orbitals are directed towards the six corners of regular octahedron. Online Chemistry tutorial IIT, CBSE Chemistry, ICSE Chemistry, engineering and medical chemistry entrance exams, Chemistry Viva, Chemistry Job interviews. Octahedral complexes have six ligands symmetrically arranged around a central atom, defining the vertices of an octahedron. The octahedron has eight faces,. Beswitched match 3rd edition.
This feature is not available right now. Please try again later. Left 4 dead 3 trailer.
Examples of octahedron in the following topics:
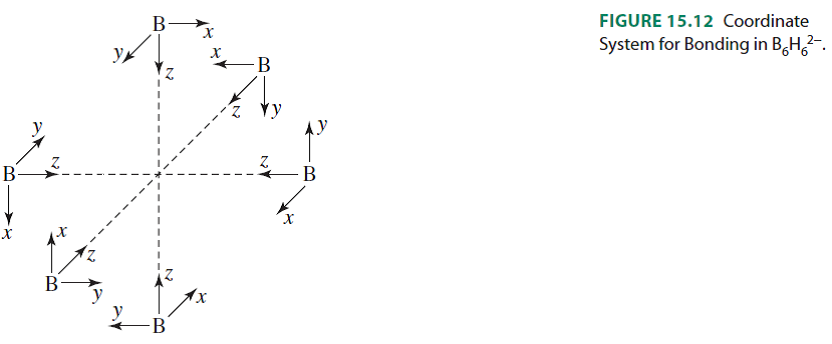
Octahedral Complexes
- Octahedral complexes have six ligands symmetrically arranged around a central atom, defining the vertices of an octahedron.
- The octahedron has eight faces, hence the prefix octa-.
Boranes: Boron-Hydrogen Compounds
- ., B5H9 an octahedron missing one vertex)
- arachno- (from the Greek for 'spiders web') B occupies n vertices of an n+2 deltahedron (e.g., B4H10 an octahedron missing two vertices)
- hypho- (from the Greek for 'net') B occupies n vertices of an n+3 deltahedron (e.g., possibly B8H16 has this structure, an octahedron missing three vertices)
Crystal Field Theory
- For example, in the case of an octahedron, the t2g set becomes lower in energy.
Tetrahedral and Square Planar Complexes
- The removal of a pair of ligands from the z-axis of an octahedron leaves four ligands in the x-y plane.
Are Real Dice Fair?
- A Platonic solids set of five dice; tetrahedron (four faces), cube/hexahedron (six faces), octahedron (eight faces), dodecahedron (twelve faces), and icosahedron (twenty faces).
Applying the VSEPR Model
- In an AX6 molecule, six electron pairs will try to point toward the corners of an octahedron (two square-based pyramids joined base-to-base).
- In an octahedral molecule, six electron pairs will try to point toward the corners of an octahedron.
Isomers in Coordination Compounds
- When three identical ligands occupy one face of an octahedron, the isomer is said to be facial, or fac.
Molecular Geometries
- The bond angles are all 90째, and just as four electron pairs experience minimum repulsion when they are directed toward the corners of a tetrahedron, six electron pairs try to point toward the corners of an octahedron.
Ionic Crystals
- The answer is that each of those six chloride ions sits at the center of its own octahedron, whose vertices are defined by six neighboring sodium ions.

